John M Lund Photography Inc/DigitalVision via Getty Images
Thesis
I consider Coya Therapeutics (NASDAQ:COYA) one of several high-potential neurodegenerative plays. The company was first covered by me in May of last year. At the time, it had just announced data from two open-label clinical studies in ALS and in Alzheimer’s disease [AD]. These studies were small and open-label, but consistently showed improvements or stabilization of patients treated with low-dose IL-2 [LD IL-2] or LD IL-2 + CTLA-4 Ig. Coya considers COYA 302, which is Coya’s proprietary formulation of LD IL-2 and CTLA-4 Ig packaged in a combination kit, as the more promising drug candidate. COYA 302 not only promotes anti-inflammatory regulatory T cells (Tregs) but also down regulates inflammatory macrophages.
The past year, the company’s progression was focused on financing, partnering and pipeline expansion.
Two near-term catalysts could now change the outlook of Coya. The company plans to initiate a Phase 2 trial in ALS – the lead indication for the time being – in the coming weeks. In the summer of 2024, Coya will report on a placebo-controlled randomized trial with LD IL-2 in AD.
Company
Introduction
Coya Therapeutics is a biotech company focused on the treatment of neurodegenerative diseases. Its treatment candidates, with Coya 302 as the lead drug, do so by modulating neuroinflammation. Coya’s COYA 302 target indications have expanded last year, including ALS, AD, frontotemporal dementia [FTD] and Parkinson’s disease [PD].
COYA 302 pipeline (Corporate Presentation)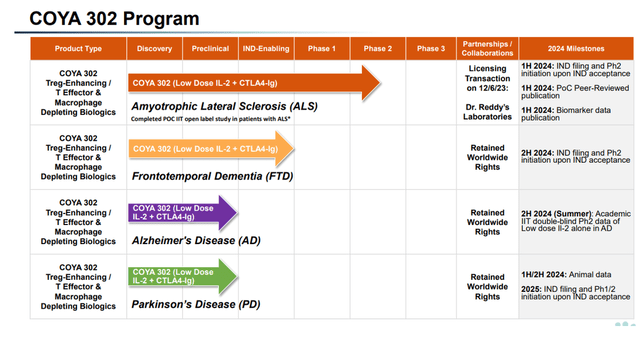
Coya wants to prioritize the development of COYA 302. Its pipeline includes Coya 301, Coya 201 and Coya 206, which I have covered before.
Coya’s pipeline looks quite different from that of a year ago, which is not atypical to biotech companies with a fairly recent IPO. At the time, programs were either discovery-stage, preclinical or IND-enabling, and AD was not yet part of the pipeline.
Each of the indications listed above is a major neurodegenerative indication with high unmet need. From that perspective, Coya appears correct to consider that COYA 302 is a pipeline within a product with >$10 billion annual sales potential, with Alzheimer’s as the largest neurodegenerative disease in the lead.
COYA 302 >10 Billion potential (Corporate Presentation)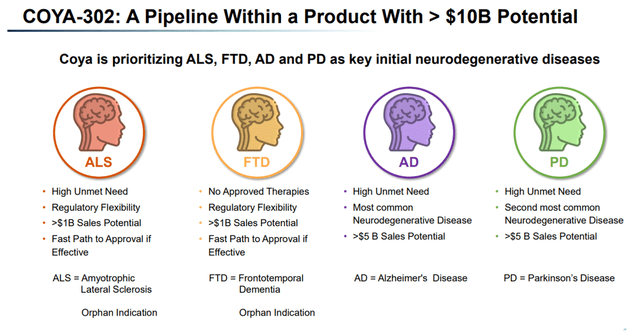
Coya’s particular anti-inflammatory approach
Modulation of the brain’s resident immune cells may be a potential treatment for neurodegenerative diseases driven by neuroinflammation, such as AD, ALS, FTD and PD. Each year, the number of drugs targeting neuroinflammation for these diseases becomes more abundant.
I believe that the way one lowers inflammation may be of critical importance; targeted reduction that allows the brain’s immune cells to perform their original functions is essential.
Regulatory T-cells or Tregs are CD4-positive T cells which are master regulators of inflammatory signaling. In the brain, they regulate immune response directly or through signaling to brain-resident immune cells such as microglia and astrocytes. Inflammation and Treg dysfunction seem to be intricately related in the brain.
Role of inflammation on neurodegeneration (Corporate Presentation)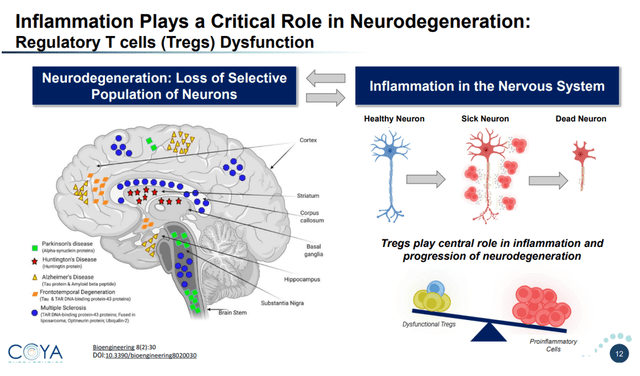
The brain-resident immune cells have two main functions. Their primary function is to nurture and sculpt the brain. Their secondary function provides an inflammatory response to foreign insults. Aggregated proteins seen in neurodegenerative diseases may also lead to pro-inflammatory signaling, including an inflammatory loop involving the brain-resident immune cells.
Complex pathophysiology of neurodegenerative diseases (Corporate Presentation)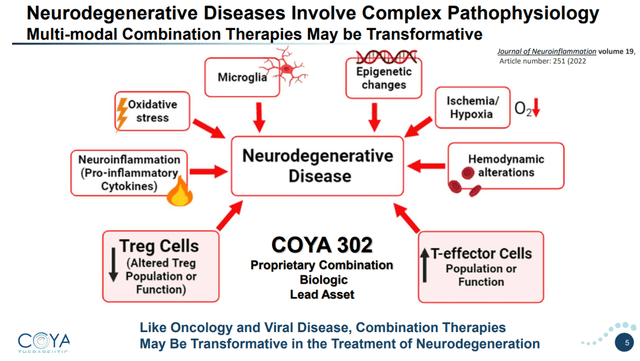
Coya’s approach for the treatment of neurodegenerative diseases does not stand alone. In 2023, there were more drugs in human trials for neuroinflammation than for any other AD-modifying factor.
Coya 301
Introduction
Coya 301 consists of proprietary subcutaneous low-dose IL-2 to upregulate Treg function and suppress pro-inflammatory T cells.
COYA 302 MoA (Corporate Presentation)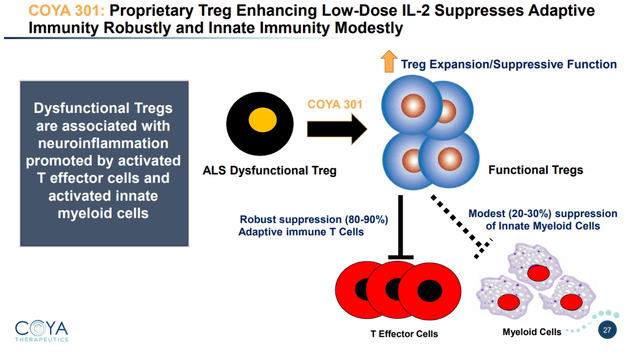
Results so far: improving cognitive function or halting disease progression
Cognition typically declines in AD patients. The mean yearly rate of decline depends on the rating scale: +4.58 points for the 7-point Adas-Cog rating scale, -4.4 points for the 30-point MMSE scale, and +1.2 points for the 18-point CDR-SB scale.
In an investigator-initiated open-label study of 8 patients with AD, four five-day monthly treatment cycles administered to patients with mild-to-moderate AD showed a statistically significant improvement in cognitive function, as measured by the MMSE. On 2 other validated scales, cognition either improved or stabilized.
COYA 301 improved or halted cognitive decline – open-label (Corporate Presentation)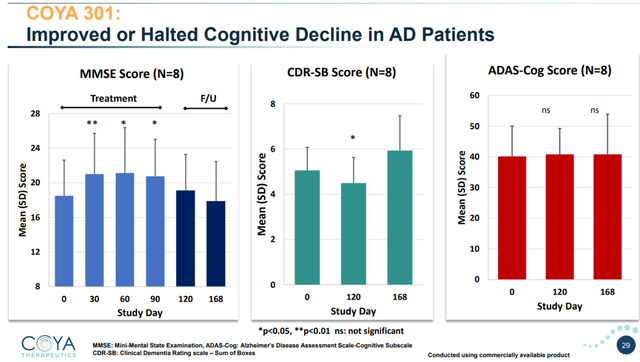
On the MMSE scale, LD IL-2 improved cognition in a statistically significant manner at 30, 60, 90 days. Reaching statistical significance in 8 patients is a major accomplishment. During the first part of the follow-up period, when no further treatment was given, cognition remained improved compared to baseline. During the second part of the follow-up period, cognition worsened, which was expected as no further treatment had been given.
On the 18-point CDR-SB scale, typical cognitive decline is about +1.2 points per year. LD IL-2 improved cognition during treatment and up to 30 days post-treatment . At that time point, the result was also statistically significant. At the end of the follow-up period, cognition had worsened as expected, as no further treatment was administered.
On the 70-point Adas-Cog scale, cognition remained stable over at 120 and 168 days.
Typically, treatment effect would be measured as a function of cognitive decline. Biogen’s (BIIB) amyloid antibody Leqembi led to 27% slowing of cognitive decline, and Eli Lilly’s (LLY) donanemab performed slightly better at 35%.
Looking at MMSE and CDR-SB here, we are seeing a halting of cognitive decline over the treatment period, with statistical significance at several time points even in this small patient population. Looking at Adas-Cog, LD IL-2 treatment also showed arrest of cognitive decline. Regardless of how one looks at this, if these results would be able to be repeated in a larger, randomized, placebo-controlled trial, we are looking at massive outperformance of anti-amyloid antibodies.
From a biomarker standpoint, LD IL-2 restored Treg function and numbers and lowered pro-inflammatory cytokines.
COYA 301 lowers inflammatory cytokines slide 1 (Corporate Presentation) COYA 301 lowers inflammatory cytokines slide 2 (Corporate Presentation)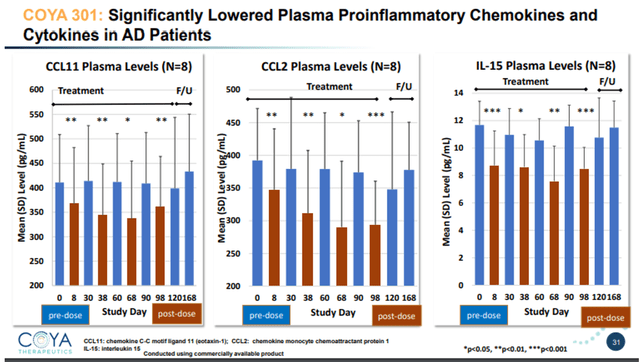
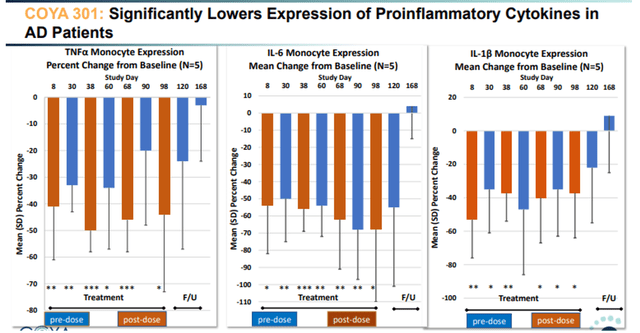
TNF-levels reductions post-dosing were between -40% and -50% compared to baseline. TNF is relevant because it is a master pro-inflammatory cytokine, and also the area of interest of INmune Bio. In conclusion, LD IL-2 showed very encouraging positive results here both on biomarker and on cognitive assessments.
Coya 302
Introduction
COYA 302, comprised of COYA 301 and CTLA-4 Ig, has a dual immunomodulatory MoA intended to enhance the anti-inflammatory function of Tregs and suppress the inflammation produced by activated monocytes and macrophages.
COYA 302 MoA (Corporate Presentation) COYA 302 biologic activity (Corporate Presentation)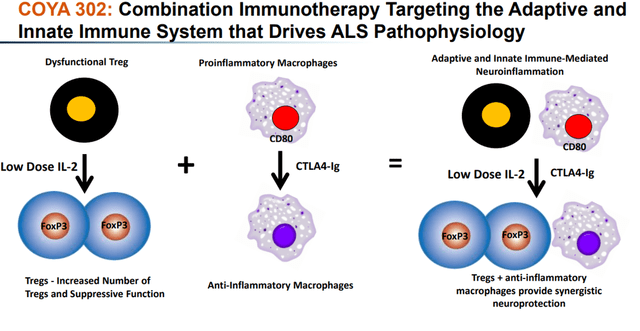
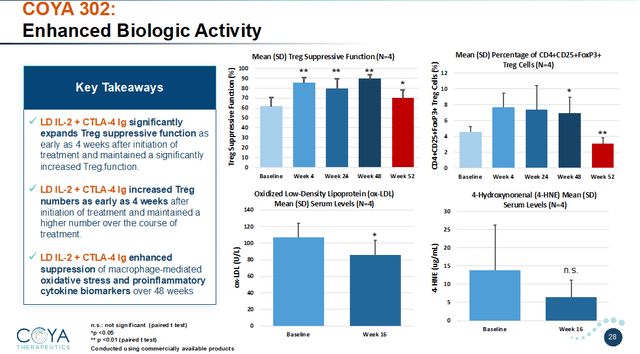
Results so far: stabilization or substantial slowdown of disease progression
LD IL-2 + CTLA-4 Ig was first administered in an open-label study in 4 patients with ALS, for 48 weeks. Coya announced results of this study in February 2023. Results on the ALSFRS-R scale, assessing motor function, were the most interesting. Patients typically decline 1 point per month on the 48-point ALSFRS-R scale. In this open-label trial, Coya’s patients were progressing at a similar rate prior to treatment.
Overall, these patients remained stable over the first 24 weeks. They only declined by 1 point for the subsequent 24 weeks. That represents a minimal change compared to the expected 12-point decline. LD IL-2 + CTLA-4 Ig either stabilized or dramatically slowed disease progression. That is totally unprecedented in ALS. During the 8-week follow-up period, when no further treatment was given, patients declined at their previous rate of 1 point per month, again proving treatment effect.
COYA 302 appears to ameliorate disease progression (Corporate Presentation)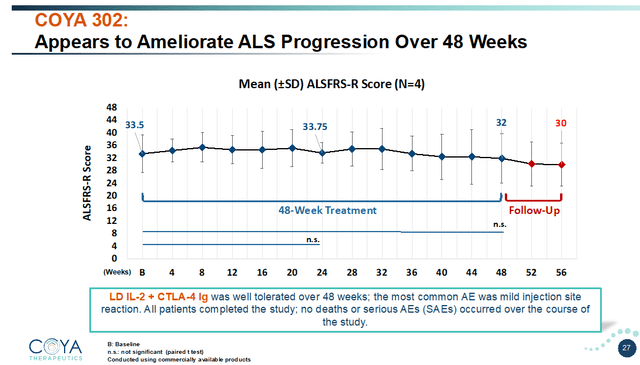
What makes this compelling in favor of COYA 302 is that ALS progresses much faster compared to AD and Parkinson’s disease, and patients in the study were seeing typical disease progression before treatment.
The unmet need in ALS is high. Compared to the existing drug Radicava, which had shown about a 33% slowing of decline, that difference is major. Intravenous Radicava received FDA approval in 2017. Amylyx’s (AMLX) drug Relyvrio had been FDA-approved in 2022, but has already been taken off the market after missing primary and secondary endpoints in a Phase 3 trials.
In April 2024, Coya reported that 4-HNE levels correlated strongly with the rate of disease progression and survival. 4-HNE is a biomarker of lipid peroxidation, known to be highly toxic for neurons.
Upcoming Phase 2 trial in ALS
The initiation of Coya’s Phase 2 trial in ALS should be around the corner at this point. It was announced to start in the second half of 2024, and that second half is now coming to an end. I therefore expect this trial to initiate in the coming weeks.
In January 2024, Coya had announced having had a successful pre-IND meeting with the FDA for the development of COYA 302 for the treatment of ALS. That hopefully results in an easy validation by the FDA after the IND-filing. After Coya has filed its IND, a 30-day period starts running, during which the FDA may request additional information before initiating the trial. I assume in any case that the IND filing and subsequent trial initiation will be in the near future.
Coya’s Phase 2 trial in ALS will be in 120 patients with an ALSFRS-R score equal or greater to 35 at screening and a well-documented rate of disease progression. The study will evaluate two dosing regimens of COYA 302 versus placebo over 24 weeks, followed by a 24-week open-label extension period. The trial’s primary endpoint is change in the ALSFRS-R score compared to baseline.
COYA 302 Phase 2 ALS study design (Corporate Presentation)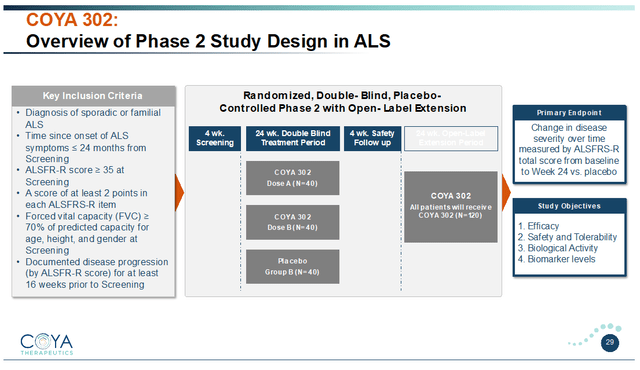
Moving into a Phase 2 trial could attract investors, and valuation generally becomes higher as a company moves into a next trial phase.
If successful, I believe this trial could give rise to an accelerated approval in ALS. Amylyx’s Relyvrio had, in fact, been approved on the basis of a randomized placebo-controlled trial in 137 patients with a six-month duration followed by an open-label extension.
For most patients with ALS, life expectancy is 2-5 years. A 24-week placebo-controlled trial followed by a -24-week open-label extension covers a large time span in which one would normally see disease progression. In comparison, the placebo decline for Radicava and Relyvrio was 7.5 and 9.97 ALSFRS-R points respectively over the course of 24 weeks.
Topline data readout in Alzheimer’s disease planned for the summer of 2024
Phase 2 randomized placebo-controlled trial in Alzheimer’s
Over the summer of 2024, Coya will report on an investigator-initiated, double-blind, placebo-controlled Phase 2 trial of LD IL-2 funded by the Bill Gates Foundation and the Alzheimer’s Association.
The 3-arm study includes 2 different dosing regimens of COYA 301 and a placebo arm. Treatments are administered over a 21-week period with a 9-week post-treatment follow-up period.
COYA 301 Phase 2 trial design (Corporate Presentation)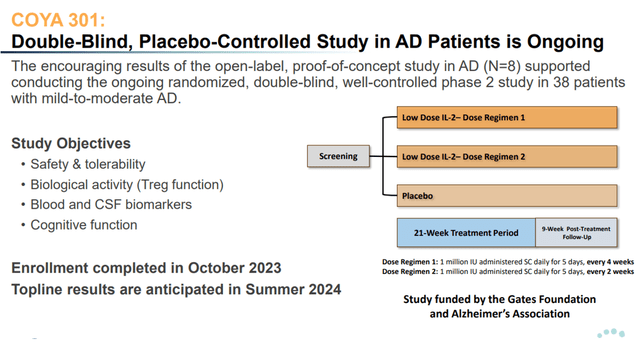
The main goal of this trial is to guide Coya towards designing a double-blind, well-controlled Phase 2 trial in AD with COYA 302. For that reason, the trial will evaluate not only safety and cognition, but a wide range of biomarkers, including NfL and GFAP. On the basis of the results, Coya can decide whether it needs to prespecify subgroups and whether it would need to include inclusion criteria into its trial, e.g. on the basis of patients’ Treg function or level of inflammation.
The thresholds for success: safety and efficacy
The recent FDA’s Advisory Committee on the Eli Lilly’s (LLY) BLA for anti-amyloid antibody donanemab has allowed the FDA’s insights into the current treatment landscape for AD and the safety issues accompanying anti-amyloid antibodies. For reference, Biogen (BIIB) has in 2024 abandoned commercialization of the much-contested anti-amyloid antibody Aduhelm. Currently, the only approved anti-amyloid antibody is Biogen/Eisai’s (OTCPK:ESALF) Leqembi, approved on an accelerated basis in January 2023 for having slowed cognitive decline by 27% over 18 months on the CDR-SB scale. The FDA had denied accelerated approval to Eli Lilly, after which Eli Lilly filed for full approval. The FDA’s concerns do not necessarily relate to efficacy, I believe, as donanemab slightly outperformed Leqembi both on its primary endpoint iADRS as on the CDR-SB rating scale.
Donanemab percentage slowing on CDR-SB (Corporate Presentation)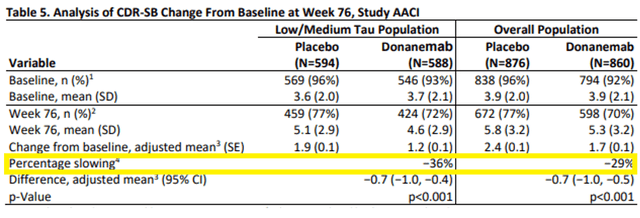
The elephant in the room is safety in my eyes. Anti-amyloid antibodies lead to so-called ARIA, brain microhemorrhage that leads to serious adverse events and death in some cases. Mortality for donanemab was substantially higher than for placebo patients, and the incidence of ARIA was quite high in patients on donanemab.
Donanemab mortality and TEAE slides (FDA briefing document donanemab AdComm)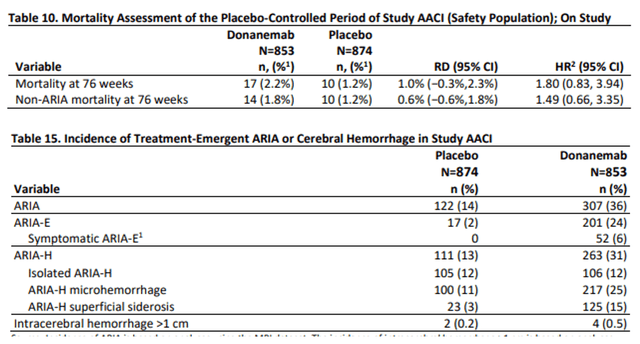
Catalyst potential
LD IL-2 is safe and well-tolerated
Contrary to anti-amyloid antibodies, LD IL-2’s safety and tolerability profile is milder. Apart from some site injection irritations, administration of LD IL-2 so far has not led to serious adverse effects. In light of previous concerns over Aduhelm as well as Leqembi, and the recent FDA’s concerns over donanemab, this accomplishment should not be underestimated.
Efficacy: potential outperformance of anti-amyloid antibodies / biomarkers
The threshold for efficacy in Alzheimer’s at this time is outperformance of anti-amyloid antibodies, slowing cognitive decline either by 27% or 29% on the CDR-SB scale. If LD IL-2 would be able to show a clean safety and tolerability record and a 30% slowing of cognitive decline, then it is a clear win and unprecedented. Never before has there been a placebo-controlled randomized trial that would have shown such safety and efficacy in Alzheimer’s disease.
If the ample biomarkers Coya is assessing in this trial would be able to underscore the effect on the neurodegenerative process, that could be a game-changer.
That, for me, is the threshold for a win at this stage.
If, additionally, this trial would show a statistically significant result, I believe that could be world news, as it could be the first placebo-controlled trial to report better results than those of the anti-amyloid antibodies in such a manner. I, however, do not count on that at this stage, given the size of the trial.
Topline data slated for summer 2024
In May 2024, Coya has announced the completion of this trial, and topline results are anticipated for the summer of 2024. Summer already starts in a week or two, so data could come faster than expected. If Coya sticks to not missing deadlines, then I believe it could be a hot summer.
Dr. Reddy’s Partnership
In December 2023, Coya announced its exclusive partnership with Dr. Reddy’s Laboratories (RDY) to develop and commercialize COYA 302 for ALS in the United States, Canada, the European Union and the United Kingdom. That partnership includes a $7.5 million upfront payment, a $4.2 million payment upon IND acceptance by the FDA, a $4.2 million payment upon first patient dosing, further regulatory milestones up to $40 million, sales-based milestone payments of up to $677.25 million linked to cumulative net sales, and royalties based on a percentage net sales of COYA 302.
I think Dr. Reddy’s has moved exceptionally soon here.
Pipeline expansion to frontotemporal dementia and Parkinson’s disease
In January 2024, Coya has announced the expansion of its pipeline for COYA 302 to FTD and PD, two other major neurodegenerative diseases with high unmet need, as all of these conditions share the same inflammatory hallmark.
Coya also plans the initiation of a Phase 2 trial in FTD, which it has now received funding from the ADDF. No trial design for this study has yet been revealed.
Strong external validation
For the microcap that Coya is, the company is remarkably well validated. I rank them in order of their importance, in my eyes.
ADDF
In May 2024, Coya announced that it had received a $5 million private placement equity investment from the Alzheimer’s Drug Discovery Foundation [ADDF]. As a leading organization, the ADDF considers controlling neuroinflammation at the forefront of new treatment ideas for AD, and expects anti-inflammatory drugs to potentially win approval over the next five years. The ADDF only funds about 25 projects per year, and typically does not take equity stakes in companies. Amounts of funding are more often below $1 million, making Coya’s $5 million funding project among ADDF’s largest in recent years.
David Einhorn
Greenlight Capital, David Einhorn’s hedge fund, is an 8.8% holder of Coya shares, up from 5% when I covered Coya a year ago. David Einhorn has been on the board of the Michael J. Fox Foundation since 2005 and should therefore be on top of the latest developments in the neurodegenerative space. I am therefore giving this equity stake extra value.
Bill and Melinda Gates Foundation / Alzheimer’s Association
Coya’s Phase 2 trial in AD is funded by the Alzheimer’s Association and the Bill and Melinda Gates Foundation.
Wilbur Ross
Former United States Secretary of Commerce Wilbur Ross, named one of the 50 most influential people in global finance by Bloomberg Markets, has joined Coya’s board in December 2023 and has recently participated in a private placement together with other institutional investors. Mr. Ross has plenty of experience in banking and private equity, and therefore is an outside validation of Coya of particular interest.
Valuation considerations
AD is the most common neurodegenerative disease in the world, affecting 5.8 million people in the US with the potential to triple by 2060, with a market size valued at $6.1 billion in 2021. Good results from the above-mentioned trial would validate the open-label data previously reported, and thereby also enhance the potential efficacy of COYA 302 for ALS, AD, PD and FTD with an estimated combined market size of $10+ billion. And these markets are growing at tremendous speeds.
At the time of writing, Coya’s market cap is $105 million. The potential for gains is huge here if results are good, whether or not they are statistically significant given the small patient population. For reference, the excitement surrounding Cassava Sciences’ (SAVA) open-label data from a small AD patient population in 2021 had seen their market cap surge to just below $5 billion. In 2021 and 2022 respectively, Evaluate saw Biogen’s Aduhelm and Eli Lilly’s donanemab respectively as the years’ most anticipated drugs, worth $4.8 and $6 billion in annual sales respectively. Eli Lilly’s stock had added more than $20 billion on the announcement of donanemab’s efficacy data, and similarly, Biogen’s stock had added more than $10 billion upon efficacy data from lecanemab.
COYA currently ranks at number 11 when it comes to performance of 2022 IPO’s, with a 40% gain. It has ranked much higher some months ago. I believe that, with good topline data, it could rank among – or above – the top performers.
Risks
Coya is a microcap biotech company. Microcap stocks are often characterized by high beta. Biotech companies can move tremendously on news.
Furthermore, regulatory risk is always present. The FDA can halt trials before their commencement, or as they are ongoing, for various reasons and not only those related to safety.
There is a large competitive risk as well. Coya is the only company I am aware of that is investigating modulation of Tregs and macrophages for the treatment of neurodegenerative diseases, but it is not the only company pursuing treatments for any of the mentioned neurodegenerative diseases. There is notably more competition in the AD space, and particularly from biotech companies such as INmune Bio (INMB), Cassava Sciences, Denali Therapeutics (DNLI), Alector (ALEC) and many others.
Financials
Coya had $36 million in cash and cash equivalents at the end of March 2024 and has picked up $24.1 million in net proceeds in a private placement some months before. Upon IND-acceptance and Phase 2 trial initiation in ALS, Coya should be eligible for payment of $4.2 million each time, totaling $8.4 million. The ADDF’s recent $5 million equity investment means further money on the bank for Coya.
The average net loss for the past two reported quarters was $3.9 million. Annual cash burn has been estimated at $11.7 million. At any of those rates, taking into account the expected Dr. Reddy’s payments but also a higher quarterly expense due to the ongoing trial, I expect Coya to be funded well past its ALS study readout.
That would not preclude the company from picking up some further financing to run a Phase 2 trial in AD, if the market would respond positively to the upcoming topline data. It is equally not excluded that at that time, a big pharma partners and facilitates the funding of further trials.
Conclusion
Coya may be at a transformative point of its progression, as the company will soon initiate a potentially registrational Phase 2 trial with COYA 302 in ALS, and will report topline data from a placebo-controlled randomized Phase 2 trial with LD IL-2 in AD this summer.
LD IL-2 and LD IL-2 + CTLA-4 Ig have yielded exceptional results in previously reported small open-label studies. Though such studies are generally not what attracts big pharma, Coya has already concluded a deal with Dr. Reddy’s for the development of COYA 302 for ALS. Further outside validation for Coya’s programs appears strong, from investors, scientists, and leading institutions.
A randomized, double-blind, placebo-controlled trial is of great value to big pharma and investors. Success is outperformance of anti-amyloid antibodies on safety and efficacy. A good safety profile and a 30% slowing of cognitive decline are sufficient. Disease stabilization or improvement would be a tremendous success, but it is not necessary. If, additionally, COYA 301 would be able to do so in this small trial in a statistically significant manner, I believe the world could be watching. The market reaction to any of the above events could be tremendous, as history has shown. To that extent, I believe Coya is very much under the radar for the moment.
Similarly, I expect Coya to initiate its potentially registrational Phase 2 trial in ALS this year. The company announced it for 2H 2024. Again, success is outperformance of Radicava’s results, which were strongly outperformed in the open-label study. If results exceed that and would be able to show disease stabilization even if for a while, the significance of that trial should not be underestimated.
For the reasons above, I am reiterating my Strong Buy rating on Coya Therapeutics.
Credit: Source link