magicmine/iStock via Getty Images
Today, we put Verona Pharma plc (NASDAQ:VRNA) back in the spotlight. We last looked at this London-headquartered biopharma in mid-summer of last year after the stock has had a strong rally. The shares have since pilfered away most of those gains. However, the company has a pivotal FDA decision on the horizon next month, making this a good time to peak back in on Verona. An updated analysis follows below.
Seeking Alpha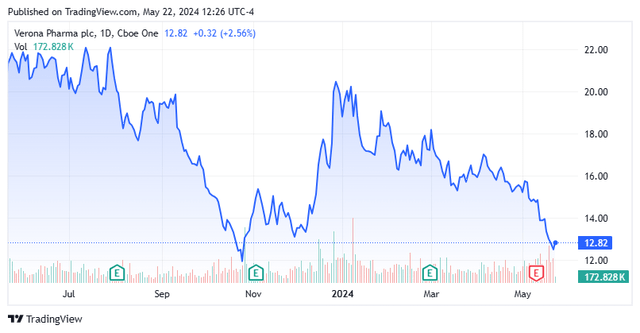
Verona Pharma’s stock currently trades around $12.50 a share and has an approximate market capitalization of just north of $1 billion. This hopefully soon to be commercial biopharma concern is targeting the developing therapies for the treatment of respiratory diseases. The company’s main asset product candidate is called Ensifentrine, which combines a bronchodilator and anti-inflammatory properties in one compound. Ensifentrine was developed to treat chronic obstructive pulmonary disease or COPD and other respiratory diseases.
February 2024 Company Presentation
There are numerous products on the market to treat COPD it should be noted. Ensifentrine is a dual inhibitor of PDE3 and PDE4 and the company is positioning it as a “first-in-class” maintenance treatment for COPD.
February 2024 Company Presentation
Verona conducted two Phase 3 studies ENHANCE-1 and ENHANCE-2 that produced positive trial data and the company submitted the new drug application for Ensifentrine early in the summer of 2023.
February 2024 Company Presentation
The company also evaluating Ensifentrine to potentially treat other indications as well. Verona is planning to kick off two Phase 2 studies in the second half of this year. One positioning Ensifentrine as part of combination therapy as a maintenance treatment of patients with COPD via delivery in a nebulizer. The other trial will evaluate Ensifentrine as a monotherapy to treat patients with non-cystic fibrosis bronchiectasis.
Recent News/Key Inflection Point:
The scheduled date for the decision on the NDA is June 26th, which is just five weeks away. Management has potentially positioned and plans to market Ensifentrine as the first new product to treat COPD in over a decade.
It should be noted that Sanofi (SFY) and Regeneron Pharmaceuticals (REGN) are in the process of trying their blockbuster drug Dupixent, an interleukin-4 and interleukin-13 inhibitor, as a potential new treatment for COPD. Dupixent has an advantage of already being approved for several other indications, as well as having a large and established sales team in place. The companies have a supplemental Biologic License Application or sBLA accepted with an action date, ironically a day after Ensifentrine’s NDA scheduled decision date on June 27th. Dupixent did over $11.5 billion in global sales in 2023 it should be noted.
In early May, the company secured a new financing agreement from OMERS Life Sciences and Oaktree Capital Management. This includes a debt facility of $400 million that will be available in five tranches. This replaced a $400 million debt facility Verona had with other lenders. Up to $250 million in additional financing can be accessed via $250M in funding can come from the sale of a redeemable interest in future Ensifentrine-related revenue.
Analyst Commentary & Balance Sheet:
The analyst community is universally enthusiastic about Verona’s prospects heading into June’s key decision date. Since March, six analyst firms, including Wedbush, Piper Sandler and BTIG have reissued Buy/Outperform ratings on the stock. Price targets proffered range from $32 to $36 a share.
According to the 10-Q filed for the first quarter, at the end of March, Verona had nearly $255 million in cash and marketable securities on its balance sheet and a term loan amount of just under $50 million. With the new debt facility secured, management has stated that Verona has funding in place now to facilitate the company’s growth through all planned commercialization beyond 2026. Verona burned through approximately $17 million in cash to support all operational activities in the first quarter.
Conclusion:
February 2024 Company Presentation
February 2024 Company Presentation
Ensifentrine is very likely to be approved late in June based on trial results, and it is aiming at a large and growing market. If the NDA is not approved, it will likely be via complete response letter related to chemistry, manufacturing and controls and not trial data. This would delay approval but could be addressed in time. Verona has also secured necessary funding to support commercialization, and the company has an initial sales team and rollout plan in place.
February 2024 Company Presentation
The $64,000 question is, giving this is a mass market with several established competitors, is what kind of sales traction will Ensifentrine garner? My experience with companies marketing a product for the first time in their history to a mass market, is sales traction is challenging in the initial stages of the rollout. Therefore, at best I would take a small “starter” position in Verona Pharma plc stock and accumulate a larger holding over time provided the company executes well against its commercialization plan. An article on Seeking Alpha in February also made the bull and bear case around VRNA and is worth reading for any investors considering this stock for investment.
Credit: Source link